Green fluorescent protein (GFP) is widely used as biomarkers and its importance is demonstrated by the three 2008 Nobel Chemistry Prize winners Osamu Shimomura, Martin Chalfie, and Roger Y. Tsien. They discovered GFP and applied to biology, which contributes enormously to understanding of the function of cells and the mechanism of diseases.
The fluorescence quantum yield of GFP is as high as 79%, but the corresponding chromophore (GFPc), p-HBDI, is unexpectedly nonfluorescent with a quantum yield lower than 0.1%. This has prompted chemists to study the excited decay mechanism of p-HBDI and to develop many GFPc-like chromophores. Over the past years, the fluorescence quenching is mainly attributed to the cis-trans isomerization reaction.
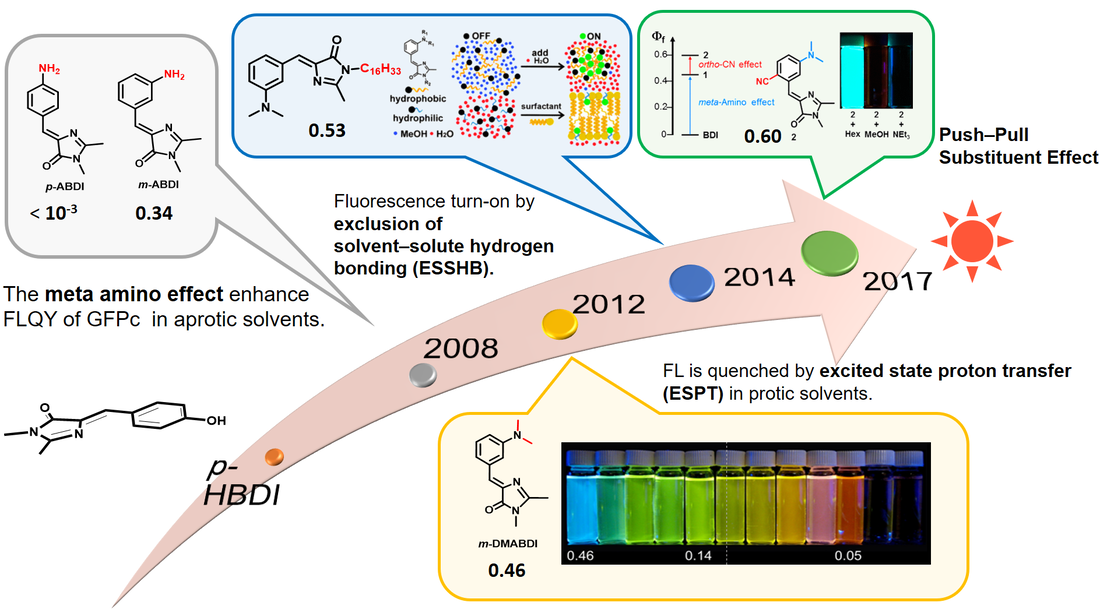
Our lab focuses on the photophysical and photochemical properties of highly fluorescent meta-amino-substituted GFPc. The following figure shows the development history of GFPc derivatives in our lab. We have successfully increased the fluorescence quantum yield from nearly 0 (p-HBDI, wild-type GFPc) to 46% (m-DMABDI). We have explored the GFPc analogues in several aspects, including hydrogen-bonding-induced fluorescence quenching, aggregation-induced fluorescence enhancement, fluorescence quantum yield enhancement, real-time cell imaging, colorful fluorescence patterning, and data encryption and decryption.
The fluorescence quantum yield of GFP is as high as 79%, but the corresponding chromophore (GFPc), p-HBDI, is unexpectedly nonfluorescent with a quantum yield lower than 0.1%. This has prompted chemists to study the excited decay mechanism of p-HBDI and to develop many GFPc-like chromophores. Over the past years, the fluorescence quenching is mainly attributed to the cis-trans isomerization reaction.
Our lab focuses on the photophysical and photochemical properties of highly fluorescent meta-amino-substituted GFPc. The following figure shows the development history of GFPc derivatives in our lab. We have successfully increased the fluorescence quantum yield from nearly 0 (p-HBDI, wild-type GFPc) to 46% (m-DMABDI). We have explored the GFPc analogues in several aspects, including hydrogen-bonding-induced fluorescence quenching, aggregation-induced fluorescence enhancement, fluorescence quantum yield enhancement, real-time cell imaging, colorful fluorescence patterning, and data encryption and decryption.
- Yang, J.-S.; Huang, G.-J.; Liu, Y.-H.; Peng, S.-M. "Photoisomerization of the Green Fluorescence Protein Chromophore and the meta-and para-Amino Analogues" Chem. Commun. 2008, 0, 1344-1346.
- Huang, G.-J.; Ho, J.-H.; Ch. Prabhakar, Liu, Y.-H.; Peng, S.-M.; Yang, J.-S. "Site-Selective Hydrogen Bonding-Induced Fluorescence Quenching of Highly Solvatofluorochromic GFP-like Chromophores" Org. Lett. 2012 14, 5034-5037.
- Tou, S.-L.; Huang, G.-J.; Chen, P.-C.; Chang, H.-T.; Tsai, J.-Y.; Yang, J.-S. "Aggregation-Induced Emission of GFP-like Chromophores via Exclusion of Solvent-Solute Hydrogen Bonding" Chem. Commun. 2014, 50, 620-622.
- Tsai, M.-S.; Ou, C.-L.; Tsai, C.-J.; Huang, Y.-C., Cheng, Y.-C.; Sun, S.-S.; Yang, J.-S. "Fluorescence Enhancement of Unconstrained GFP Chromophore Analogs Based on the Push-Pull Substituent Effect" J. Org. Chem. 2017, 82, 8031-8039.