Pentiptycene has a non-planar rigid backbone which might induce different packing modes in the solid state, and thus is an excellent scaffold for the design of mechanochromic luminescent materials. Also, because of the cavities created by U- and V-shape notches, which are suitable for host-guest interaction, pentiptycene has potential for sensing application. Our recently published papers below give a preliminary but typical illustration.
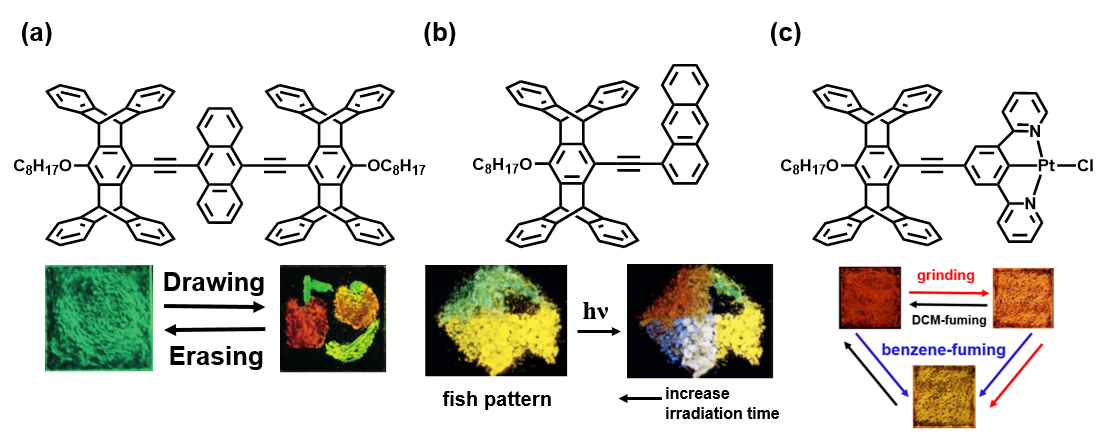
In the first example, we combine pentiptycene and anthracene to construct the molecule in figure (a). In the solid state, it can form exciplex with different anilines, providing a fluorescence change from green to red. Mechanical grinding also decreases its reactivity to guest molecules and thus a memory effect can be achieved. Like the fruit painting below, we unprecedentedly present five colors (red, orange, yellow, green and black) at the same time.
The second example is shown in figure (b). The pristine and ground powders of the molecule have yellow and blue fluorescence, respectively. Controlling the extent of grinding, we can obtain near-pure white emission. It also has two polymorphs, the yellow and green emissive single crystals. Under UV irradiation, the products formed by the photochemical reaction cause stress in the bulk crystal, which turns the fluorescence into blue and red respectively. This special phenomenon is termed as photomechanochromism. The fluorochromicity is demonstrated by the angelfish patterning.
We also utilize pentiptycene in metallorganic system to form the platinum complex in figure (c). After the molecule is ground or fumed by benzene vapor, its packing can be altered and the phosphorescence interconverts among red, orange and yellow. Such benzene-responsive Pt complexes are rarely reported in literature.
Our lab is dedicated to developing pentiptycene derivatives. We hope to achieve more colorful luminescence change and more diversified regulation to expand the application of these smart materials.
In the first example, we combine pentiptycene and anthracene to construct the molecule in figure (a). In the solid state, it can form exciplex with different anilines, providing a fluorescence change from green to red. Mechanical grinding also decreases its reactivity to guest molecules and thus a memory effect can be achieved. Like the fruit painting below, we unprecedentedly present five colors (red, orange, yellow, green and black) at the same time.
The second example is shown in figure (b). The pristine and ground powders of the molecule have yellow and blue fluorescence, respectively. Controlling the extent of grinding, we can obtain near-pure white emission. It also has two polymorphs, the yellow and green emissive single crystals. Under UV irradiation, the products formed by the photochemical reaction cause stress in the bulk crystal, which turns the fluorescence into blue and red respectively. This special phenomenon is termed as photomechanochromism. The fluorochromicity is demonstrated by the angelfish patterning.
We also utilize pentiptycene in metallorganic system to form the platinum complex in figure (c). After the molecule is ground or fumed by benzene vapor, its packing can be altered and the phosphorescence interconverts among red, orange and yellow. Such benzene-responsive Pt complexes are rarely reported in literature.
Our lab is dedicated to developing pentiptycene derivatives. We hope to achieve more colorful luminescence change and more diversified regulation to expand the application of these smart materials.
- Matsunaga, Y.; Yang, J.-S. "Multicolor Fluorescence Writing Based on Host-Guest Interactions and Force-Induced Fluorescence-Color Memory" Angew. Chem. Int. Ed. 2015, 54, 7985-7989.
- Hsu, L.-Y.; Maity, S.; Matsunaga, Y.; Hsu, Y.-F.; Liu, Y.-H.; Peng, S.-M.; Shinmyozu, T.; Yang, J.-S. "Photomechanochromic vs Mechanochromic Fluorescence of a Unichromophoric Bimodal Molecular Solid: Multicolor Fluorescence Patterning" Chem. Sci. 2018, DOI:10.1039/C8SC03740J.
- Lin, C.-J.; Liu, Y.-H.; Peng, S.-M.; Shinmyozu, T.; Yang, J.-S. "Excimer-Monomer Photoluminescence Mechanochromism and Vapochromism of Pentiptycene-Containing Cyclometalated Platinum(II) Complexes" Inorg. Chem. 2017, 56, 4978-4989.