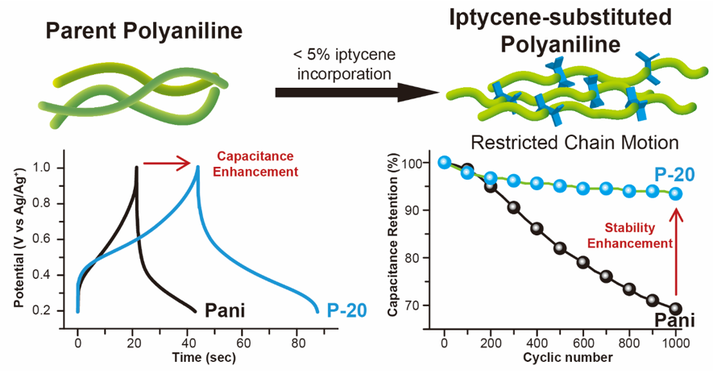
The increased energy demand has prompted the search of not only renewable energy but also efficient energy storage devices. Because of the merits of high power density, fast charge/discharge ability, and long cycle life, supercapacitors have emerged as promising energy storage devices. To achieve high charge capacity, the nature of the electrodes should be of high conductivity, surface area, and electrochemical stability. Among the various conducting materials for applications as the electrode materials in supercapacitors, conducting polymers such as polyaniline (PANI) have the advantages of low cost and low environmental impacts. However, the electrochemical stability is generally a limitation of conducting polymers. To overcome this problem, we have incorporated the rigid and nonplanar pentiptycene scaffolds into PANI to improve the electrochemical stability and meanwhile to enhance the specific capacitance. By restricting the chain motions of polyanilines, the stability after 1000 consecutive charging-discharging cycles are highly enhanced. An evidence on SEM study shows that the degree of contour blurring and volume change of the nanogranules are relatively minor for the pentiptycene-substituted polyaniline. Furthermore, the accompanied enhancement in capacitance and conductivity might indicate that the clipping of polymer chains facilitates interchain π-π stacking interactions and thus interchain conductivity. Further studies on enhancing specific capacitance and capacitance retention are in progress.
- Tan, W. S.; Lin, S.-Y.; Yang, J.-S. "Synthesis, Structural Characterization, and Electrochemical Properties of Isotruxene–Polyaniline Hybrid Systems." J. Chin. Chem. Soc. 2017, 64 (9), 1007-1022.
- Tan, W. S.; Lee, T.-Y.; Hsu, Y.-F.; Huang, S.-J.; Yang, J.-S. "Iptycene Substitution Enhances the Electrochemical Activity and Stability of Polyanilines." Chem Comm. 2018, 54 (43), 5470-5473.